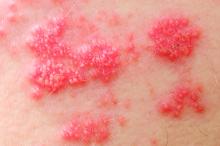
Among patients with moderate to severe ulcerative colitis, a median of 1.4 years and up to 4.4 years of tofacitinib therapy was safe apart from a dose-related increase in risk of herpes zoster infection, according to an integrated analysis of data from five clinical trials.
Compared with placebo, a 5-mg twice-daily maintenance dose of tofacitinib (Xeljanz) produced a 2.1-fold greater risk of herpes zoster infection (95% confidence interval, 0.4-6.0), while a 10-mg, twice-daily dose produced a statistically significant 6.6-fold increase in incidence (95% CI, 3.2-12.2).
With the exception of the higher incidence rate of herpes zoster, “in the overall cohort, the safety profile of tofacitinib was generally similar to that of tumor necrosis factor inhibitor therapies,” wrote William J. Sandborn, MD, director of the inflammatory bowel disease center and professor of medicine, at the University of California, San Diego, and associates. The findings were published in Clinical Gastroenterology and Hepatology.
Tofacitinib is an oral, small-molecular Janus kinase inhibitor approved in the United States for treating moderate to severe ulcerative colitis, as well as rheumatoid and psoriatic arthritis. The recommended ulcerative colitis dose is 10 mg twice daily for at least 8 weeks (induction therapy) followed by 5 or 10 mg twice daily (maintenance). The safety of tofacitinib has been studied in patients with rheumatoid arthritis through 9 years of treatment. To begin a similar undertaking in ulcerative colitis, Dr. Sandborn and associates pooled data from three 8-week, double-blind, placebo-controlled induction trials, as well as one 52-week, double-blind, placebo-controlled maintenance trial and one ongoing open-label trial. All patients received twice-daily tofacitinib (5 mg or 10 mg) or placebo.
Among 1,157 tofacitinib recipients in the pooled analysis, 84% received an average of 10 mg twice daily. For every 100 person-years of tofacitinib exposure, there were an estimated 2.0 serious infections, 1.3 opportunistic infections, 4.1 herpes zoster infections, 1.4 malignancies (including nonmelanoma skin cancer, which had an incidence of 0.7), 0.2 major adverse cardiovascular events, and 0.2 gastrointestinal perforations. The likelihood of these events did not increase with time on tofacitinib, the researchers said.